The FDA has authorized 2 novel oral antivirals for the treatment of COVID-19. The medications currently under consideration are Merck’s molnupiravir and Pfizer’s paxlovid.
In addition to Fluvoxamine which is an Antidepressant (SSRIs), it was found that it has an anti-inflammatory effect in which it binds to sigma-1 receptor immune cells thus reducing cytokines.
Novel COVID 19 Oral Antivirals:
1- Lagevrio by MSD
molnupiravir, can be used to treat adults with COVID-19 who do not require supplemental oxygen and who are at increased risk of developing severe COVID-19. Lagevrio should be administered as soon as possible after diagnosis of COVID-19 and within 5 days of the start of symptoms. The medicine, which is available as capsules, should be taken twice a day for 5 days.
2- Paxlovid by Pfizer
Nirmatrelvir tablets and Ritonavir tablets, co-packaged for oral use, it is a SARS-CoV-2 protease inhibitor antiviral therapy, specifically designed to be administered orally so that it can be prescribed at the first sign of infection or at first awareness of an exposure, potentially helping patients avoid severe illness which can lead to hospitalization and death. PF-07321332 is designed to block the activity of the SARS-CoV-2-3CL protease, an enzyme that the coronavirus needs to replicate. Co-administration with a low dose of ritonavir helps slow the metabolism, or breakdown, of PF-07321332 in order for it to remain active in the body for longer periods of time at higher concentrations to help combat the virus.
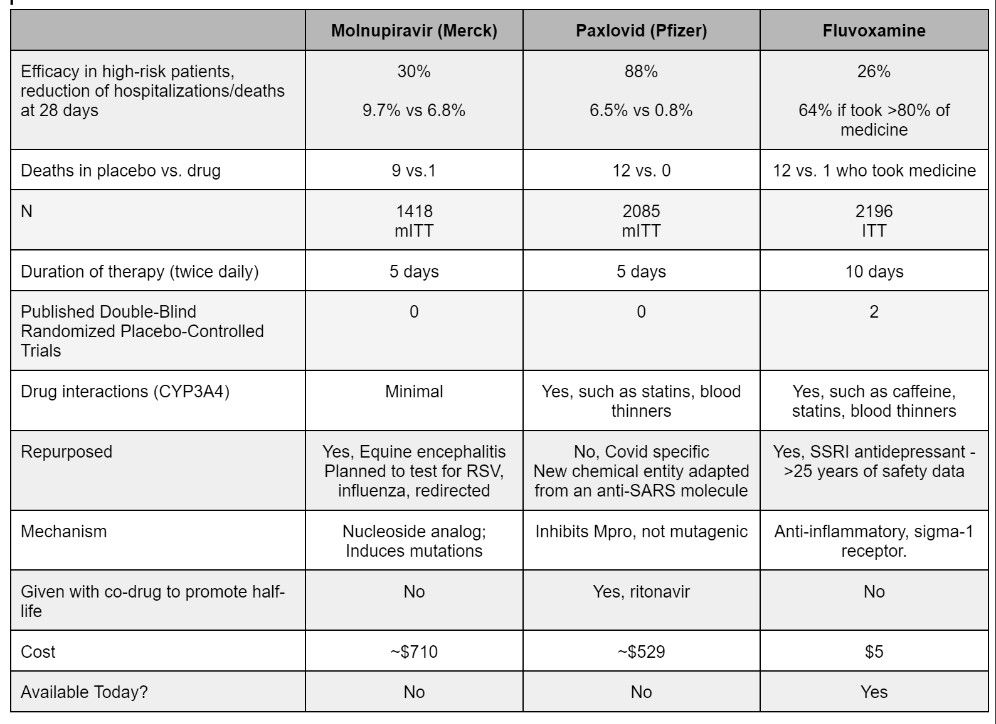
Here below is a quick comparison of both oral antiviral medications Pfizer VS MSD plus the SSRI Fluvoxamine.